Single Photon Emission Computed Tomography (SPECT): Principles, Components, Applications, and How the System Works
Single Photon Emission Computed Tomography (SPECT) is one of the most clinically powerful tools in the nuclear medicine imaging arsenal, enabling biomedical engineers and clinicians alike to visualize functional physiological processes within the human body in three dimensions. Unlike purely structural modalities such as the CT scanner or the X-ray machine, SPECT leverages gamma-emitting radiotracers to map blood flow, metabolic activity, and receptor binding with exquisite sensitivity. It complements other advanced modalities including MRI scanners and ultrasound machines by providing functional rather than purely anatomical information. Biomedical engineers working in the field of radiological devices must possess a thorough understanding of SPECT system architecture, physics, and clinical integration — particularly given its expanding role in cancer diagnosis and treatment and its regulatory landscape governed by FDA device classification frameworks. This article provides a comprehensive technical overview of SPECT scanners covering their physical principles, core components, system variants, clinical applications, regulatory considerations, maintenance practices, and future engineering trends.
Table of Contents
- What is a SPECT Scanner?
- Why is a SPECT Scanner Used?
- How Does a SPECT Scanner Work?
- Main Components of a SPECT Scanner
- Types and Variants of SPECT Scanners
- Clinical Applications of SPECT Scanners
- Regulatory and Safety Considerations
- Maintenance and Quality Assurance
- Future Trends and Engineering Innovations
1. What is a SPECT Scanner?
A SPECT scanner is a nuclear medicine imaging device that produces three-dimensional tomographic maps of the distribution of gamma-emitting radiotracers within the body. The acronym stands for Single Photon Emission Computed Tomography, distinguishing it from Positron Emission Tomography (PET), which relies on coincidence detection of annihilation photon pairs. In SPECT, individual gamma photons — single photons — emitted by the radiotracer are detected by one or more rotating gamma camera heads positioned around the patient.
The history of SPECT is rooted in the development of the Anger gamma camera by Hal Anger in the early 1960s, which initially produced planar (2D) scintigraphic images. By the 1970s, David Kuhl and other pioneers began exploring tomographic reconstruction techniques, and by the 1980s, true 3D SPECT imaging had become clinically established using filtered back-projection algorithms. The landmark development of hybrid SPECT/CT systems in the early 2000s — integrating a diagnostic CT scanner onto the same gantry — transformed SPECT from a purely functional modality into a combined anatomical-functional imaging platform, dramatically improving lesion localization and attenuation correction accuracy.
From a biomedical engineering perspective, SPECT sits at the intersection of radiation physics, detector technology, signal processing, and clinical instrumentation. It is a Class II medical device regulated by the FDA and subject to strict quality assurance protocols. Understanding SPECT at a systems engineering level requires familiarity with scintillation physics, collimator design, electronic signal chains, iterative reconstruction mathematics, and radiation safety standards — all of which are explored throughout this article.
2. Why is a SPECT Scanner Used?
SPECT is used clinically whenever physicians need to assess organ function, regional blood flow, metabolic activity, or molecular receptor distribution — information that purely structural imaging modalities simply cannot provide. While a CT scanner reveals anatomy with superb spatial resolution, it cannot distinguish viable myocardium from scar tissue, nor can it differentiate active infection from sterile inflammation. SPECT fills this critical diagnostic gap.

Data obtained from SPECT
Functional and Physiological Insight
The fundamental advantage of SPECT lies in its ability to image physiological processes in vivo at the molecular level. By labeling biologically active compounds with gamma-emitting isotopes — most commonly Technetium-99m (99mTc) with a 140 keV gamma energy and a 6-hour half-life, or Iodine-123 (123I) with a 159 keV gamma energy — clinicians can track where these tracers accumulate based on blood flow, metabolism, or receptor binding. This yields functional 3D maps of the brain, heart, skeleton, kidneys, liver, lungs, and thyroid, among other organs.
Clinical Decision-Making Value
In cardiology, SPECT myocardial perfusion imaging (MPI) is a gold-standard technique for identifying ischemic heart disease — detecting regions of reduced perfusion that indicate arterial blockage before a patient experiences a myocardial infarction. In neurology, brain SPECT studies support the diagnosis of Alzheimer’s disease, Parkinson’s disease, epilepsy, and psychiatric disorders by revealing characteristic patterns of regional hypoperfusion. In oncology, SPECT bone scans with 99mTc-labeled methylene diphosphonate (MDP) detect osseous metastases with high sensitivity, making them indispensable in staging and treatment response monitoring. These clinical utilities make SPECT a cost-effective and widely available imaging solution compared to PET, particularly in facilities without access to on-site cyclotrons.
3. How Does a SPECT Scanner Work?
The operational principle of SPECT involves four sequential stages: radiotracer administration, gamma photon emission and collimation, scintillation detection and signal processing, and 3D tomographic image reconstruction.
Radiotracer Administration and Distribution
A gamma-emitting radiotracer is administered intravenously (or in some cases orally or by inhalation) and distributes throughout the body according to its pharmacokinetic properties. For example, 99mTc-sestamibi is taken up by viable myocardial cells in proportion to blood flow, while 99mTc-HMPAO crosses the blood-brain barrier and is trapped in brain tissue proportional to cerebral perfusion. After an appropriate uptake period — typically 15 to 60 minutes depending on the tracer — the patient is positioned on the scanner table.
Gamma Camera Rotation and Data Acquisition
The gamma camera head (or heads, in multi-head configurations) rotates around the patient, acquiring 2D planar projection images — called views or projections — at angular intervals of 3° to 6° over a 360° arc (or 180° arc for cardiac studies). Each projection captures the spatial distribution of gamma photons arriving from a particular angle. The collimator restricts detection to photons traveling along defined trajectories, ensuring that spatial information is preserved. Typical acquisition times range from 15 to 30 minutes for a complete scan.
Image Reconstruction
The acquired angular projections are fed into reconstruction algorithms to generate cross-sectional tomographic slices. The classical approach is Filtered Back-Projection (FBP), which applies a ramp filter in the frequency domain to suppress low-frequency noise before back-projecting each view. While computationally straightforward, FBP is susceptible to streak artifacts, particularly in low-count studies. Modern SPECT systems predominantly employ iterative reconstruction algorithms — most notably the Ordered Subsets Expectation Maximization (OSEM) algorithm — which model the statistical nature of photon counting and incorporate corrections for attenuation, scatter, and detector response, yielding substantially superior image quality at lower radiation doses.
4. Main Components of a SPECT Scanner
A SPECT scanner is a complex electromechanical and optoelectronic system comprising several interdependent subsystems. Each component plays a critical role in determining system sensitivity, spatial resolution, energy resolution, and overall image quality.
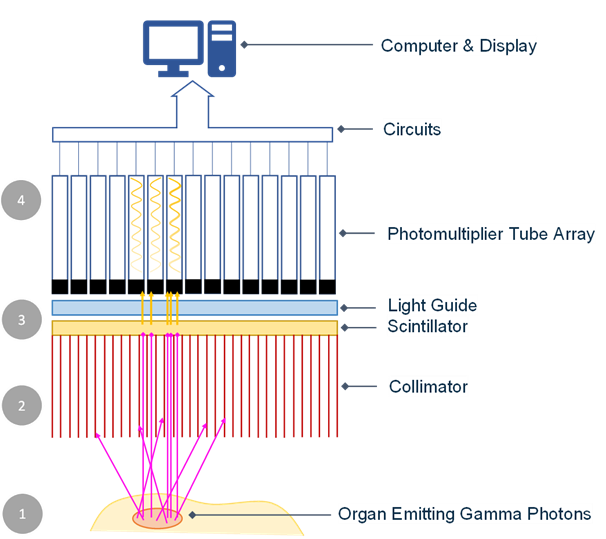
Collimator
The collimator is a lead plate perforated with thousands of parallel holes, mounted directly in front of the detector crystal. It acts as a spatial filter, permitting only gamma photons traveling parallel to the hole axis to reach the detector, thereby encoding directional (spatial) information. Parallel-hole collimators are the most common type used in clinical SPECT, available in low-energy high-resolution (LEHR), low-energy high-sensitivity (LEHS), and medium-energy configurations depending on the isotope and clinical application. The collimator is the primary determinant of system spatial resolution and sensitivity, and there is an inherent trade-off between the two: narrower holes improve resolution but reduce sensitivity. Fan-beam and cone-beam collimators offer improved sensitivity for specific organ imaging such as the brain.
NaI(Tl) Scintillation Detector
Behind the collimator lies a large-area sodium iodide crystal doped with thallium — NaI(Tl) — typically 3/8 inch (9.5 mm) thick and up to 50 × 40 cm in area per detector head. When a gamma photon is absorbed by the crystal, it produces a flash of visible light (scintillation) proportional in intensity to the photon energy. The NaI(Tl) crystal offers good energy resolution (~10% at 140 keV) and high light yield, making it well-suited for 99mTc imaging. The crystal is hermetically sealed to prevent moisture degradation, a critical engineering and maintenance consideration.
Photomultiplier Tubes (PMTs) and Signal Processing Electronics
An array of photomultiplier tubes (PMTs) — typically 60 to 100 per detector head — is optically coupled to the rear face of the crystal via a light guide. Each PMT converts the scintillation light into an electrical signal and amplifies it by a factor of 10⁶ or more through a cascade of dynodes. The Anger logic circuit processes the outputs of all PMTs simultaneously, calculating the X-Y position of each gamma interaction within the crystal using weighted centroid algorithms, and determines the energy of the interaction using a pulse height analyzer (PHA). Only events within a defined energy window (typically ±10% around the photopeak) are accepted, rejecting Compton-scattered photons that would degrade image contrast.
Gantry, Patient Table, and Reconstruction Workstation
The motorized gantry supports the detector head(s) and controls their rotation with precision stepper motors, ensuring accurate angular positioning throughout the acquisition orbit. The patient table must provide a rigid, flat, and low-attenuation surface with motorized longitudinal translation for whole-body scans. The acquisition and reconstruction workstation houses the software for scan protocol configuration, real-time data acquisition monitoring, iterative reconstruction processing, attenuation and scatter correction, and clinical display tools including multiplanar reformatting and polar map generation for cardiac studies.
5. Types and Variants of SPECT Scanners
SPECT scanner designs have diversified significantly over the decades to address specific clinical needs, optimize acquisition speed, enhance image quality, and improve patient throughput. The principal variants differ in the number of detector heads, detector material technology, and whether they are integrated with anatomical imaging modalities.
Overview of SPECT Scanner Variants
The table below summarizes the key types of SPECT scanners, their distinguishing features, and their primary clinical applications from a biomedical engineering standpoint.
| Type | Detector Heads | Detector Material | Key Feature | Primary Application |
|---|---|---|---|---|
| Single-Head SPECT | 1 | NaI(Tl) | Low cost, versatile, slower acquisition | General nuclear medicine, bone scans |
| Dual-Head SPECT | 2 | NaI(Tl) | 2× sensitivity gain, faster scans, most common clinical configuration | Whole-body bone scans, oncology, brain, cardiac |
| Triple-Head SPECT | 3 | NaI(Tl) | 3× sensitivity, optimal for brain SPECT, higher system cost | Brain perfusion, neurological studies |
| Cardiac SPECT (CZT-based) | Multiple fixed solid-state detectors | Cadmium Zinc Telluride (CZT) | No rotating gantry, superior energy resolution (~6%), 5–10× faster acquisition | Myocardial perfusion imaging (MPI) |
| Brain-Dedicated SPECT | 3 (typically) | NaI(Tl) | Small-bore design, optimized geometry for head imaging | Dementia, epilepsy, psychiatric disorders |
| Hybrid SPECT/CT | 2 (SPECT) + CT subsystem | NaI(Tl) + CT detector array | Combined functional-anatomical imaging, CT-based attenuation correction | Oncology, cardiology, infection localization |
| Digital SPECT | 2 (standard configuration) | NaI(Tl) + Silicon Photomultipliers (SiPM) | Digital light detection replaces analog PMTs, improved uniformity and energy resolution | General SPECT, evolving clinical use |
The dual-head SPECT system remains the workhorse of clinical nuclear medicine departments worldwide, offering the best balance of sensitivity, scan time, versatility, and cost. CZT-based dedicated cardiac SPECT systems have transformed myocardial perfusion imaging by enabling stress-rest studies in under 10 minutes with substantially reduced radiation dose — a significant advantage over conventional NaI(Tl) systems. Hybrid SPECT/CT configurations, analogous in concept to PET/CT platforms, have become the standard of care in most modern facilities by enabling precise anatomical co-registration and eliminating photon attenuation artifacts that historically plagued standalone SPECT studies. The emerging digital SPECT platforms incorporating silicon photomultiplier technology promise further improvements in count rate linearity, spatial uniformity, and system stability, representing the next engineering frontier in SPECT instrumentation.
6. Clinical Benefits and Diagnostic Advantages of SPECT
Functional and Metabolic Imaging Capability
The most clinically significant advantage of SPECT imaging over purely anatomical modalities is its ability to capture functional and metabolic processes in vivo. Rather than depicting structural morphology alone, SPECT maps physiological parameters such as regional myocardial perfusion, cerebral blood flow, bone remodeling activity, and receptor binding density. This makes it an indispensable tool in cardiology, neurology, and oncology, where pathology often manifests at the functional level before any detectable structural change. For biomedical engineers working in clinical imaging departments, understanding this distinction is critical when evaluating device performance requirements and selecting appropriate radiotracers in consultation with nuclear medicine physicians. As explored in our overview of radiological devices for biomedical engineers, functional imaging modalities occupy a unique niche that structural imaging simply cannot replicate.
Non-Invasive Whole-Body Assessment and Hybrid Imaging
SPECT is a non-invasive technique that enables whole-body surveys with a single radiotracer administration, making it particularly valuable for staging metastatic disease, evaluating multifocal skeletal involvement, and assessing systemic conditions. The advent of SPECT/CT hybrid systems has further elevated diagnostic confidence by co-registering functional scintigraphic data with high-resolution anatomical CT images. Systems such as the Siemens Healthineers Symbia Intevo and GE Healthcare Discovery NM/CT exemplify this synergy, allowing precise anatomical localization of tracer uptake abnormalities that would otherwise be ambiguous on planar or SPECT images alone. Biomedical engineers should appreciate that SPECT/CT integration introduces additional engineering considerations including mechanical alignment calibration, CT-based attenuation correction pipelines, and radiation dose management across both modalities. Compared to standalone CT scanners, the hybrid configuration demands coordinated quality assurance protocols that span both detector systems.
Wide Availability and Clinical Versatility
Compared to PET imaging, SPECT benefits from broader global availability due to its use of longer-lived radiotracers that do not require an on-site cyclotron. Technetium-99m, the most widely used SPECT tracer with a 6-hour half-life, is readily produced from molybdenum-99/technetium-99m generators available at most nuclear medicine departments. This logistical advantage translates to lower operational costs and greater accessibility in both developed and resource-limited healthcare settings. The clinical applications span myocardial perfusion imaging (MPI), bone scintigraphy, thyroid and parathyroid imaging, renal function studies, sentinel lymph node mapping, and neurological assessments including dopaminergic system evaluation with DaTscan. This versatility means that a single SPECT system can serve multiple clinical departments, delivering strong return on investment for hospital procurement teams.
7. Risks, Limitations, and Engineering Challenges
Radiation Dose and Patient Safety Considerations
SPECT involves the administration of radiopharmaceuticals, exposing patients to ionizing radiation with effective doses typically ranging from 5 to 20 mSv depending on the tracer used, administered activity, and imaging protocol. For reference, a standard Tc-99m myocardial perfusion study delivers approximately 9–12 mSv, which is comparable to several years of natural background radiation exposure. Biomedical engineers must work closely with medical physicists and nuclear medicine specialists to implement dose optimization strategies, including the use of weight-based dosing protocols, stress-only imaging protocols in MPI, and modern iterative reconstruction algorithms that permit activity reduction without compromising image quality. Regulatory frameworks such as IEC 60601-1 and FDA 510(k) requirements mandate that radiation safety features be integral to device design and deployment. For a broader discussion of how devices are regulated and classified, the article on FDA biomedical device classification provides essential context.
Spatial Resolution Limitations and Collimator Trade-offs
A fundamental physical limitation of SPECT is its spatial resolution, which typically ranges from 8 to 15 mm full-width at half-maximum (FWHM) under clinical conditions — significantly inferior to the sub-millimeter resolution achievable with modern CT or MRI scanners. This resolution is fundamentally constrained by the collimator, a lead structure placed in front of the detector crystal that physically restricts which gamma photons are accepted based on their angle of incidence. The collimator introduces an inherent sensitivity-resolution trade-off: high-resolution collimators improve spatial definition but drastically reduce count sensitivity, resulting in noisier images or longer acquisition times. Parallel-hole, fan-beam, and pinhole collimator geometries each optimize different aspects of this trade-off for specific clinical applications. Biomedical engineers involved in system procurement or upgrade decisions must evaluate collimator options carefully against the department’s primary clinical workflow.
Acquisition Time, Motion Artifacts, and Radiopharmacy Dependencies
Standard SPECT acquisitions require 15 to 30 minutes of patient imaging time, during which even minor patient motion can introduce significant reconstruction artifacts, including blurring, misalignment between angular projections, and false perfusion defects in cardiac studies. Motion correction algorithms and gating techniques — such as ECG-gated SPECT for cardiac applications — partially mitigate this issue but add complexity to both acquisition protocols and post-processing workflows. Additionally, SPECT depends entirely on a functioning radiopharmacy supply chain, including generator availability, sterile compounding facilities, and strict quality control of radiopharmaceutical preparations. Disruptions in the global Mo-99 supply chain, as experienced during past nuclear reactor outages, can halt SPECT services entirely. Biomedical engineers should advocate for contingency planning and build familiarity with radiopharmacy operations as part of their broader clinical engineering responsibilities.
8. Recent Technological Innovations in SPECT
CZT Solid-State Detectors and Digital SPECT
The transition from conventional sodium iodide [NaI(Tl)] scintillation crystals coupled to photomultiplier tubes (PMTs) toward cadmium zinc telluride (CZT) solid-state semiconductor detectors represents the most transformative hardware advancement in SPECT technology in recent decades. CZT detectors convert gamma photons directly into electrical signals without an intermediate scintillation step, yielding superior energy resolution (approximately 5–6% at 140 keV versus 9–10% for NaI/PMT systems), improved intrinsic spatial resolution, and faster timing characteristics. These properties enable more effective scatter rejection, simultaneous dual-isotope imaging, and significant reductions in administered activity while maintaining diagnostic image quality. Purpose-built CZT cardiac cameras such as the GE Healthcare NM 530c and the Spectrum Dynamics D-SPECT employ stationary multi-detector ring or focused detector geometries that eliminate detector rotation, dramatically reducing scan times from 15–30 minutes to as little as 2–4 minutes for myocardial perfusion studies. This innovation directly addresses patient throughput constraints and is particularly valuable for hemodynamically unstable or claustrophobic patients.
AI-Driven Reconstruction and Quantitative SPECT
Artificial intelligence and deep learning are rapidly reshaping SPECT image reconstruction and post-processing workflows. Convolutional neural network (CNN)-based reconstruction algorithms can suppress image noise, reduce streak artifacts, and enhance apparent spatial resolution without increasing radiation dose — effectively decoupling the traditional noise-dose trade-off. Commercial implementations are already appearing in platforms such as the Siemens Healthineers Symbia Intevo with xSPECT technology, which leverages CT-derived anatomical boundaries to constrain iterative reconstruction and achieve resolution recovery approaching 4–6 mm. Simultaneously, the field is progressing toward fully quantitative SPECT, in which pixel values are expressed as absolute activity concentrations (MBq/mL) rather than relative counts. Quantitative SPECT is enabling dosimetry-guided radionuclide therapy, particularly for Lu-177 DOTATATE treatments in neuroendocrine tumors, representing a critical intersection of diagnostic and therapeutic nuclear medicine. Biomedical engineers with expertise in signal processing and software validation play a central role in deploying and verifying these AI-based tools, as discussed in our article on top skills every biomedical engineer should master. The role of advanced imaging in oncology is also explored in our feature on how biomedical engineering is advancing cancer diagnosis and treatment.
9. Key Takeaways for Biomedical Engineers
Quality Control, Calibration, and Regulatory Compliance
Biomedical engineers responsible for SPECT systems must implement rigorous quality control (QC) programs aligned with NEMA NU 1 standards and manufacturer specifications. Daily QC procedures include flood field uniformity testing using a Co-57 sheet source or Tc-99m flood phantom to detect detector non-uniformities, PMT gain drifts, or crystal damage. Weekly and monthly protocols extend to sensitivity calibration, energy resolution verification, spatial resolution assessment using bar phantoms, and center-of-rotation (COR) corrections critical for artifact-free tomographic reconstruction. For SPECT/CT hybrid systems, CT-to-SPECT registration accuracy must be validated periodically. Compliance with IEC 60601-2-65 (gamma camera performance), IEC 60601-1 (general electrical safety), ISO 13485 (quality management systems), and applicable FDA 510(k) clearance documentation must be maintained and audited. Engineers should maintain detailed QC logbooks and trend analysis dashboards to detect performance degradation before it affects clinical diagnoses.
Preventive Maintenance, PACS Integration, and Radiopharmacy Coordination
Preventive maintenance schedules for SPECT systems should encompass PMT or CZT detector module inspection, gantry motion system lubrication and encoder calibration, high-voltage power supply verification, and cooling system performance checks. Biomedical engineers should coordinate with manufacturers’ field service engineers on annual planned maintenance visits and maintain a parts inventory for high-failure-rate components such as PMT assemblies, collimator latching mechanisms, and detector shielding integrity. PACS and RIS integration requires validation of DICOM conformance statements, particularly for nuclear medicine DICOM objects (DICOM SC, NM Image Storage SOP classes), and ensuring correct patient demographic mapping to avoid study misattribution. Collaboration with the nuclear medicine technologist team and radiopharmacist is essential for synchronized workflow: the biomedical engineer must understand radiopharmaceutical preparation timelines, dose calibrator calibration requirements, and the impact of generator elution schedules on imaging capacity. This multidisciplinary competency is a hallmark of effective clinical engineering practice, as highlighted throughout our coverage of radiological device management.
Attenuation Correction and Image Reconstruction Oversight
Attenuation correction (AC) is a critical post-processing step in SPECT that compensates for the depth-dependent attenuation of gamma photons traversing body tissues, which otherwise introduces artifactual defects — most notably inferior wall artifacts in cardiac SPECT. CT-based AC in SPECT/CT systems requires accurate co-registration between the CT transmission map and SPECT emission data; respiratory motion mismatch between the breath-hold CT and free-breathing SPECT can introduce misregistration artifacts that mimic pathology. Biomedical engineers should be knowledgeable about iterative reconstruction algorithms such as OSEM (Ordered Subsets Expectation Maximization) and its variants, understanding how the number of iterations and subsets influence noise-resolution trade-offs. Oversight of AI-assisted reconstruction tool validation, including phantom-based performance benchmarking and ongoing clinical audit against established diagnostic criteria, falls squarely within the biomedical engineer’s scope of technical responsibility. Staying current with evolving reconstruction standards and vendor software updates is essential for maintaining image quality and regulatory compliance.
References
- Slomka PJ, Pan T, Germano G. Recent Advances and Future Progress in PET Instrumentation. Semin Nucl Med. 2016;46(1):5-19. PubMed PMID: 29330249.
- Bocher M, Blevis IM, Tsukerman L, et al. A fast cardiac gamma camera with dynamic SPECT capabilities: design, system validation and future potential. Eur J Nucl Med Mol Imaging. 2010;37(10):1887-1902. PubMed PMID: 31806873.
- Shiri I, Arabi H, Salimi Y, et al. Deep-learning-based image reconstruction and post-processing in nuclear medicine. Eur J Nucl Med Mol Imaging. 2021;48(13):4073-4076. PubMed PMID: 33494119.
- International Electrotechnical Commission. IEC 60601-1: Medical Electrical Equipment – Part 1: General Requirements for Basic Safety and Essential Performance. Geneva: IEC; 2005 (consolidated edition 2012).
- International Electrotechnical Commission. IEC 60601-2-65: Medical Electrical Equipment – Part 2-65: Particular Requirements for the Basic Safety and Essential Performance of Gamma Cameras. Geneva: IEC; 2012.
- National Electrical Manufacturers Association. NEMA NU 1-2018: Performance Measurements of Gamma Cameras. Rosslyn, VA: NEMA; 2018.
- International Organization for Standardization. ISO 13485:2016 Medical Devices – Quality Management Systems – Requirements for Regulatory Purposes. Geneva: ISO; 2016.
- Siemens Healthineers. Symbia Intevo SPECT/CT System – Product Overview. Erlangen: Siemens Healthineers; 2023. Available at: siemens-healthineers.com.
- GE Healthcare. Discovery NM/CT and NM 530c SPECT Systems – Product Information. Chicago: GE Healthcare; 2023. Available at: gehealthcare.com.
- Spectrum Dynamics Medical. D-SPECT Cardiac Imaging System – Technical Overview. Caesarea: Spectrum Dynamics; 2023. Available at: spectrumdy.com.
- U.S. Food and Drug Administration. 510(k) Premarket Notification Database – Nuclear Medicine Gamma Camera Clearances. Silver Spring: FDA; 2024. Available at: accessdata.fda.gov.
- Cherry SR, Sorenson JA, Phelps ME. Physics in Nuclear Medicine. 4th ed. Philadelphia: Elsevier Saunders; 2012.
