a comprehensive overview of CT scanners for biomedical engineers: physics, components, types, clinical applications, risks an the latest innovations in computed tomography
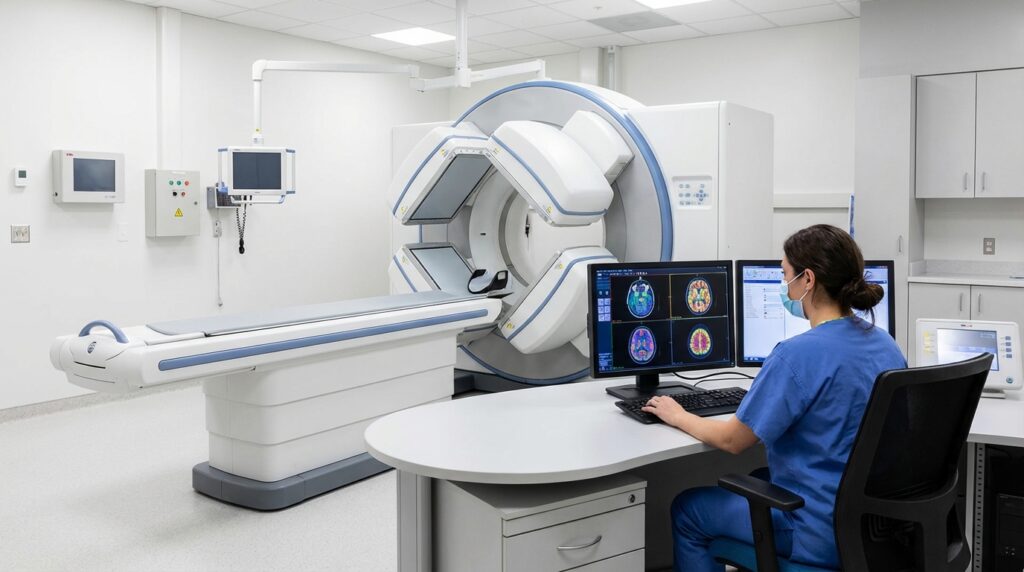
The CT scanner (Computed Tomography scanner) stands as one of the most transformative diagnostic instruments in the history of modern medicine, fundamentally reshaping how clinicians visualize internal anatomy and make critical diagnostic decisions. For biomedical engineers, understanding the physics, engineering design, and clinical utility of CT systems is essential to supporting procurement, quality assurance, maintenance, and optimization of these devices within complex hospital environments. As a key member of the broader family of radiological devices, the CT scanner complements modalities such as ultrasound machines and magnetic resonance imaging to deliver comprehensive diagnostic coverage across virtually every clinical specialty.
- What is a CT Scanner?
- Why is a CT Scanner Used?
- How Does a CT Scanner Work?
- What Are the Main Components of a CT Scanner?
- What Types and Variants of CT Scanners Exist?
- What Are the Main Benefits of a CT Scanner?
- What Are the Risks and Limitations of CT Scanners?
- How Is the CT Scanner Evolving? Recent Innovations
- Key Takeaways and Tips for Biomedical Engineers
What is a CT Scanner?
Definition and Historical Origins
A computed tomography (CT) scanner is a sophisticated medical imaging device that uses a rotating X-ray source and an array of detectors to acquire multiple cross-sectional projection images of the human body. Unlike a conventional X-ray machine, which superimposes all anatomical structures onto a single two-dimensional projection, CT reconstructs a fully three-dimensional volumetric dataset, enabling clinicians and biomedical engineers to visualize internal anatomy with extraordinary spatial and contrast resolution. CT is classified among the broader family of radiological devices and represents one of the most clinically significant inventions in modern diagnostic medicine.
The technology’s origins trace directly to the work of British electrical engineer Sir Godfrey Newbold Hounsfield at EMI Laboratories. Beginning in 1967, Hounsfield conceptualized a method by which X-ray transmission measurements taken at many angles around an object could be mathematically combined to reconstruct internal structure without physical dissection. After years of development, the world’s first clinical CT scan was performed on October 1, 1971, at Atkinson Morley Hospital in Wimbledon, London, producing an image of a cerebral cyst in a conscious patient. This landmark event transformed neurological diagnosis overnight. Concurrently, mathematician Allan Cormack had independently developed the mathematical framework for reconstruction, and both Hounsfield and Cormack were jointly awarded the Nobel Prize in Physiology or Medicine in 1979, cementing CT’s place in medical history. Within a decade of that first clinical scan, CT had expanded from head-only units to whole-body systems capable of imaging every organ system.
The Hounsfield Unit Scale
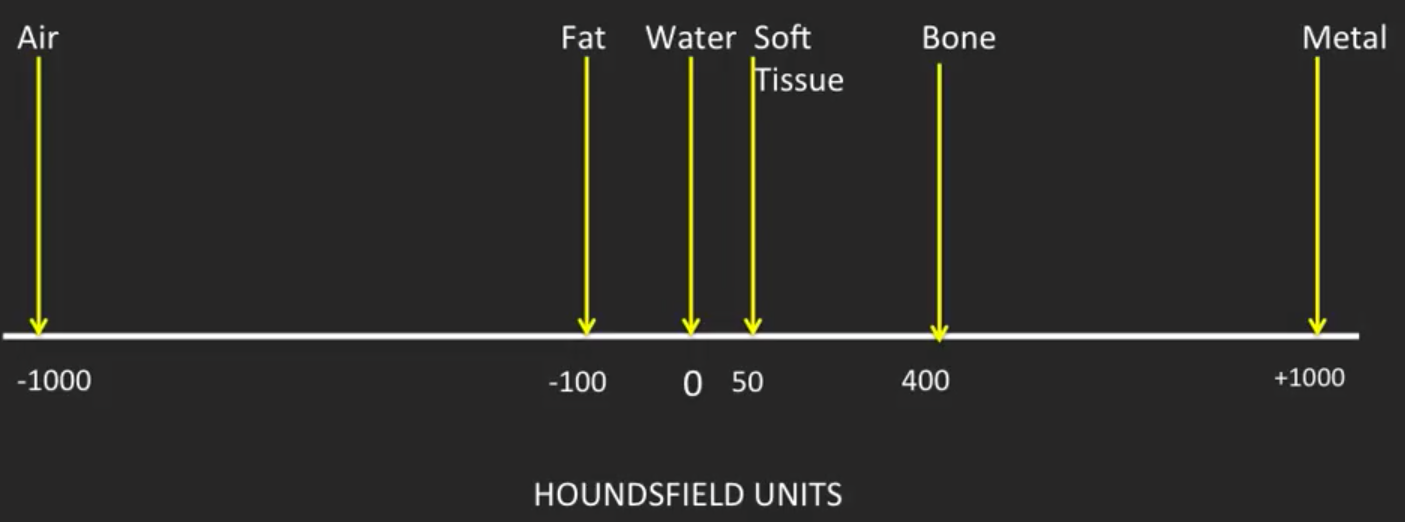
Central to CT imaging is the Hounsfield Unit (HU) scale, a quantitative measure of X-ray linear attenuation coefficient normalized relative to water. By definition, distilled water at standard temperature and pressure is assigned an HU value of 0, while air is assigned −1000 HU. At the other extreme, dense cortical bone typically registers between +400 and +1000 HU, and metallic implants can exceed +3000 HU. Soft tissues occupy the intermediate range: fat typically falls between −100 and −50 HU, solid organs such as the liver range from +50 to +70 HU, and clotted blood may reach +70 to +90 HU. This linear, calibrated scale allows radiologists and engineers to perform tissue characterization, differentiate pathological from normal tissue, and apply threshold-based segmentation algorithms for three-dimensional rendering. The quantitative nature of the HU scale is a fundamental advantage over plain projection radiography, where only gross opacity differences are visible, and it also distinguishes CT from modalities such as ultrasound machines, which rely on acoustic impedance rather than photon attenuation.
Why is a CT Scanner Used?
Clinical Indications
CT scanning encompasses an extraordinarily broad range of clinical applications across virtually every medical specialty. In oncology, CT is the workhorse of tumor staging, surveillance, and treatment response assessment, providing reproducible volumetric measurements of lesions within the chest, abdomen, and pelvis. In neurology, non-contrast CT of the brain remains the fastest and most accessible method for detecting acute hemorrhagic stroke, intracranial hypertension, and traumatic brain injury, where rapid diagnosis is critical for patient survival. CT angiography of the coronary arteries, known as coronary CT angiography (CCTA), has emerged as a powerful non-invasive tool for evaluating coronary artery disease, offering submillimeter spatial resolution of calcified and non-calcified plaques without catheterization. Pulmonary CT angiography is the gold standard for diagnosing pulmonary embolism, replacing ventilation-perfusion scintigraphy in most centers. In musculoskeletal radiology, CT excels at characterizing complex fractures, cortical bone destruction in osteomyelitis, and joint pathology, supplementing the soft-tissue sensitivity of MRI. It also plays a complementary role to the DEXA scanner in quantifying bone mineral density through opportunistic screening protocols.
Role in Emergency and Critical Care
In emergency medicine and critical care, CT has become indispensable for rapid triage of polytrauma patients. A whole-body trauma CT, sometimes called a pan-scan, can survey the head, cervical spine, chest, abdomen, and pelvis within seconds, identifying life-threatening injuries such as solid organ lacerations, pneumothorax, aortic injury, and pelvic ring fractures in a single acquisition. The speed advantage of CT over MRI is decisive in these contexts; while an MRI examination may require 30 to 60 minutes, a modern 64-slice or higher CT scanner can complete a diagnostic study in under 10 seconds of actual scan time. This temporal efficiency, combined with the broad availability of CT in emergency departments globally, makes it the preferred cross-sectional modality when minutes determine outcome. In intensive care units, portable or mobile CT systems extend diagnostic imaging to patients too unstable for transport to the radiology department, further expanding the device’s clinical reach.
How Does a CT Scanner Work?
X-Ray Attenuation and Projection Data
The physical foundation of CT imaging is the Beer-Lambert law of X-ray attenuation, which states that the intensity of a monoenergetic X-ray beam decreases exponentially as it passes through matter: I = I₀ · e^(−μx), where I₀ is the incident intensity, μ is the linear attenuation coefficient of the material, and x is the path length. In a polychromatic clinical CT beam, the measured signal at each detector element represents the line integral of attenuation along each ray path through the patient. By acquiring thousands of such line integrals at many angular positions around the patient, the scanner accumulates a complete sinogram — a two-dimensional map of projection data — from which the cross-sectional attenuation map (i.e., the CT image) can be mathematically reconstructed. Each detector reading is digitized and transmitted to the data acquisition system for processing.
Scanner Generations
CT technology has evolved through several distinct engineering generations. First-generation scanners, including Hounsfield’s prototype, used a single pencil beam and a single detector in a translate-rotate geometry, requiring several minutes per slice. Second-generation systems introduced a narrow fan beam with multiple detectors, reducing scan time. Third-generation scanners, which form the basis of nearly all modern clinical systems, use a wide fan beam rotating continuously with a detector arc that spans the entire patient diameter, eliminating translational motion and enabling sub-second rotation times. Fourth-generation designs placed detectors in a full stationary ring with only the X-ray tube rotating, but this approach proved less efficient and is rarely used today. The introduction of slip ring technology in the late 1980s enabled continuous gantry rotation without cable wrap-around, which in turn made helical (spiral) scanning possible — a paradigm shift that allowed volumetric acquisition of entire body regions in a single breath-hold.

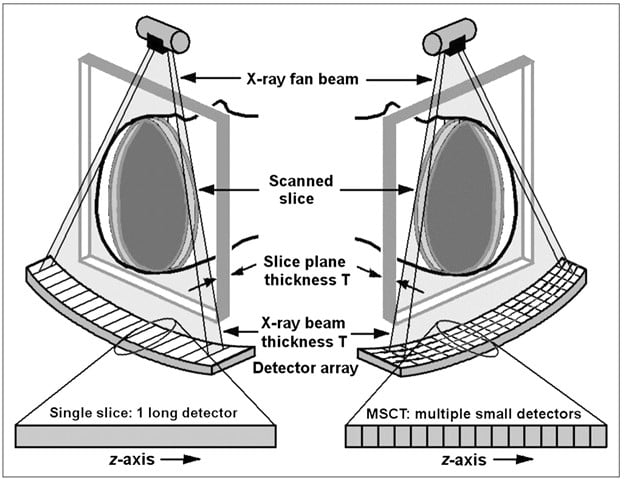
Image Reconstruction Algorithms
Image reconstruction converts raw projection data into diagnostic images. The classical algorithm, filtered back-projection (FBP), applies a ramp filter to the projection data to correct for the blurring inherent in simple back-projection, then smears each filtered projection back across the image matrix. FBP is computationally fast and deterministic but amplifies quantum noise at low-dose settings. Iterative reconstruction (IR) algorithms, including model-based iterative reconstruction (MBIR) and vendor-specific methods such as ASIR (GE), SAFIRE (Siemens), and iDose (Philips), incorporate statistical noise models and system optics into an optimization loop that alternately projects estimated images forward and compares them to measured data, progressively refining the solution. Iterative techniques can achieve equivalent diagnostic image quality at 30–60% lower radiation dose compared to FBP. More recently, deep learning reconstruction (DLR) algorithms trained on large paired datasets have demonstrated the ability to denoise and sharpen images with superior noise texture and lesion detectability at ultra-low doses, representing the current frontier of CT image processing.
What Are the Main Components of a CT Scanner?
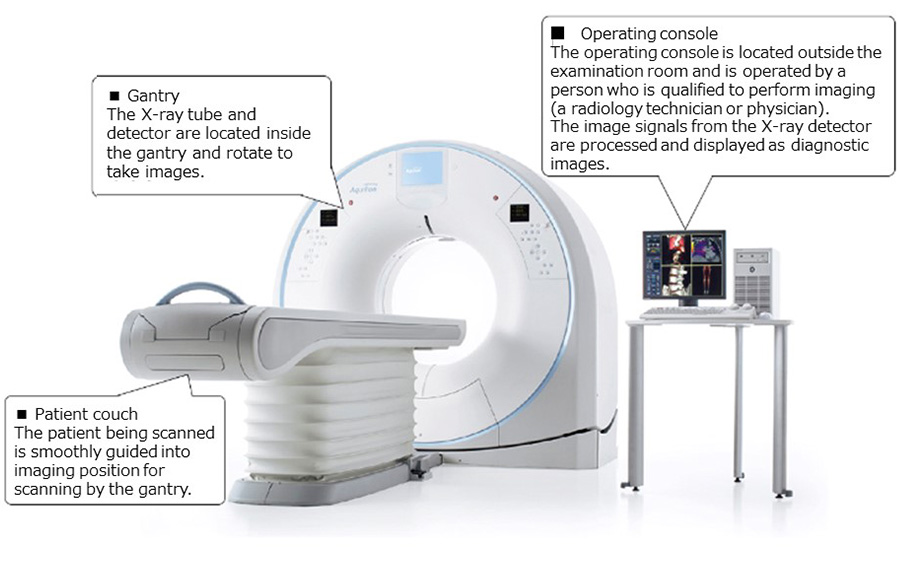
X-Ray Tube and High-Voltage Generator
The X-ray tube is the most mechanically and thermally demanding component of the CT system. Modern CT tubes are rotating-anode designs in which electrons emitted from a heated tungsten cathode are accelerated across a potential difference of typically 80 to 140 kilovolts and directed onto a rotating tungsten-rhenium alloy anode disk spinning at 9,000 to 10,000 RPM. The rotating anode distributes heat over a larger focal track area, enabling the sustained high-power outputs required for rapid volumetric scanning — often 50 to 100 kilowatts continuously. Heat management is critical; modern tubes incorporate graphite backing on the anode for thermal storage, liquid metal bearing systems for frictionless high-speed rotation, and sophisticated anode cooling mechanisms. The high-voltage generator must deliver precisely regulated, ripple-free kilovoltage to ensure consistent beam quality and accurate HU calibration throughout the rotation.
Detector Array
Contemporary CT detectors are solid-state scintillator arrays, replacing the older xenon gas ionization chambers used in earlier generations. Each detector element consists of a scintillator crystal, typically gadolinium oxysulfide (GOS) or cadmium tungstate, optically coupled to a photodiode. When X-ray photons strike the scintillator, visible light photons are generated and converted to an electrical signal proportional to X-ray intensity. Modern multi-detector CT (MDCT) arrays contain thousands of individual detector elements arranged in multiple rows along the z-axis (patient length direction), enabling simultaneous acquisition of 64, 128, 256, or even 320 slices per rotation. The geometric efficiency, dynamic range, and afterglow characteristics of the detector material directly influence image quality and dose performance.
Gantry and Slip Ring Technology
The gantry is the large toroidal housing that contains and rotates the X-ray tube, detector array, and associated electronics. Modern gantry apertures are typically 70 to 90 centimeters in diameter to accommodate bariatric patients and interventional positioning. Gantry rotation speeds have improved from 1 second per revolution in early CT systems to as fast as 0.24 seconds per revolution in current high-performance scanners, dramatically improving temporal resolution for cardiac imaging. Slip ring technology — a system of conductive rings and brushes or contactless electromagnetic coupling — enables continuous electrical power and signal transfer to and from the rotating components without the need for cables, which would otherwise limit rotation to a single turn. This engineering breakthrough was the essential enabler of helical scanning and, subsequently, of multi-detector CT as we know it today.
Data Acquisition System and Computer
The data acquisition system (DAS) digitizes the analog signals from each detector channel with high precision and at extremely high sampling rates, typically generating several gigabits of raw data per second during a scan. The DAS feeds preprocessed projection data to a high-performance reconstruction computer equipped with multiple graphics processing units (GPUs) or application-specific integrated circuits (ASICs) capable of reconstructing high-resolution volumetric images within seconds of scan completion. Reconstructed image data are transferred via high-speed networks to the hospital’s Picture Archiving and Communication System (PACS), where they are stored in DICOM format and made accessible to clinicians at workstations throughout the institution. Proper FDA device classification and cybersecurity compliance for the networked DAS and PACS interface are important regulatory considerations for biomedical engineers managing CT infrastructure.
What Types and Variants of CT Scanners Exist?
From Single-Slice to Ultra-High-Resolution Systems
The evolution of CT scanner technology has been defined largely by the progressive increase in the number of simultaneously acquired detector rows, a parameter that directly governs scan speed, z-axis coverage, and slice thickness. Single-slice CT (SSCT), the technology standard through the early 1990s, acquired one axial image per gantry rotation. The introduction of 4-slice MDCT in 1998 quadrupled throughput and enabled routine abdominal CT in a single breath-hold. Subsequent generations of 16-slice, 64-slice, 128-slice, and 256/320-slice systems progressively expanded z-coverage per rotation from a few centimeters to as much as 16 centimeters in a single revolution, allowing organs such as the heart, liver, and entire brain to be imaged in one or two rotations. Dual-source CT scanners, introduced by Siemens in 2005, mount two X-ray tube–detector pairs offset by approximately 90 degrees within the same gantry, effectively halving the temporal resolution to below 75 milliseconds — sufficient to freeze cardiac motion at elevated heart rates without beta-blocker premedication.
Specialized CT Variants
Beyond the mainstream clinical scanner, several specialized CT variants address specific diagnostic niches. Dual-energy CT (DECT) acquires data at two different X-ray spectra — either simultaneously using dual-source hardware, rapid kilovoltage switching, or dual-layer detector technology — enabling material decomposition into basis substance maps (e.g., iodine, calcium, uric acid), virtual monoenergetic image synthesis, and virtual non-contrast generation, all from a single acquisition. Cone beam CT (CBCT) uses a flat-panel detector and a diverging cone-shaped beam to acquire volumetric data with very high spatial resolution at low cost, and is widely deployed in dentomaxillofacial imaging, orthopedic surgery suites, and radiation therapy treatment rooms. Portable and mobile CT systems, mounted on wheeled platforms or dedicated vehicles, bring cross-sectional imaging to neurocritical care units, operating theaters, and remote or austere environments. PET-CT combines positron emission tomography metabolic imaging with CT anatomical localization in a single integrated gantry, and remains the backbone of oncologic staging and treatment response assessment worldwide. The table below summarizes the key characteristics of each CT type for biomedical engineering reference.
| Type | Slices/Rotation | Scan Speed | Key Application | Relative Dose |
|---|---|---|---|---|
| Single-slice CT | 1 | Slow (1 slice/rotation) | Early head/body imaging | High (older technology) |
| 4-slice MDCT | 4 | Moderate | Routine body CT, abdomen | Moderate |
| 16-slice MDCT | 16 | Fast | Trauma, vascular CT | Moderate |
| 64-slice MDCT | 64 | Very fast | Cardiac CT angiography, oncology | Low-moderate |
| 256/320-slice MDCT | 256–320 | Extremely fast | Whole-organ single-rotation, dynamic perfusion | Low (wide coverage, fewer rotations) |
| Dual-source CT | 64–192 (per source) | Ultra-fast temporal resolution (<75 ms) | High-heart-rate cardiac imaging, pediatric | Low |
| Dual-energy CT | 64–192 | Fast (single acquisition) | Gout, renal stones, iodine mapping, oncology | Low-moderate |
| Cone Beam CT | Variable (flat-panel) | Moderate | Dental, ENT, orthopedic, radiation therapy | Very low (for dental CBCT) |
| Portable CT | 8–32 | Moderate | ICU neuro, intraoperative, remote settings | Moderate |
What Are the Main Benefits of a CT Scanner?
Diagnostic Accuracy, Speed, and Clinical Versatility
CT scanners have fundamentally transformed diagnostic medicine by offering a unique combination of speed, resolution, and volumetric coverage that no other single imaging modality can match in acute clinical settings. Modern multi-detector CT (MDCT) systems can image the entire thorax or abdomen in under five seconds, making them indispensable in emergency and trauma care where rapid triage decisions directly determine patient outcomes. The high spatial resolution achievable with current 64-slice and beyond systems — often reaching sub-millimeter isotropic voxels — allows clinicians to detect lesions, fractures, vascular abnormalities, and organ pathology with exceptional anatomical fidelity. The standardized Hounsfield Unit (HU) scale ensures consistent tissue characterization across different scanner platforms, enabling radiologists to reliably distinguish soft tissue, fat, fluid, calcification, and hemorrhage without ambiguity.
The versatility of CT extends across virtually every clinical specialty. In emergency medicine, CT is the gold standard for diagnosing traumatic brain injury, pulmonary embolism, aortic dissection, and intra-abdominal hemorrhage. In oncology, it plays a central role in tumor detection, staging, and treatment response monitoring, often complemented by metabolic data from PET-CT systems. Cardiologists rely on cardiac CT angiography (CCTA) for noninvasive coronary artery assessment, while orthopedic surgeons depend on high-resolution bone imaging and 3D reconstructions for complex fracture evaluation. Pulmonologists utilize high-resolution CT (HRCT) for interstitial lung disease characterization, and urologists employ CT urography for calculi and urothelial lesion assessment. This breadth of application means that a single CT installation serves multiple departments, maximizing return on institutional investment. For a broader perspective on the range of radiological tools in biomedical practice, see Radiological Devices: All You Must Know as a Biomedical Engineer.
Role in Surgical Planning and Treatment Monitoring
Beyond initial diagnosis, CT scanning is integral to preoperative planning across surgical disciplines. Three-dimensional volume rendering reconstructions derived from thin-slice CT acquisitions allow surgeons to virtually rehearse complex procedures involving craniofacial reconstruction, hepatic resection, renal surgery, and spinal instrumentation. CT-guided biopsy and ablation procedures have further expanded the interventional role of the technology, enabling minimally invasive tissue sampling and thermal ablation of tumors in the liver, kidney, and lung with real-time image guidance. Intraoperative CT systems, including O-arm platforms, provide surgeons with immediate feedback on implant placement during orthopedic and neurosurgical procedures, reducing revision rates and improving safety.
In radiation oncology, CT-based treatment planning is the cornerstone of conformal radiotherapy, stereotactic radiosurgery, and proton beam therapy. Simulation CT scans define the gross tumor volume (GTV) and organs at risk (OAR), which inform the dose prescription and beam geometry. Serial CT imaging during chemotherapy or targeted therapy cycles provides objective response assessment according to standardized RECIST (Response Evaluation Criteria in Solid Tumors) criteria, allowing oncologists to modify treatment regimens based on measurable anatomical changes. This iterative, evidence-based approach to treatment monitoring is a hallmark of modern precision oncology. For more on how biomedical engineering drives advances in cancer diagnosis and treatment, visit How Biomedical Engineering Is Advancing Cancer Diagnosis and Treatment.
CT Versus MRI and Conventional X-Ray: Comparative Advantages
When compared to conventional projection radiography, CT provides cross-sectional and volumetric data that eliminates the superimposition of overlying structures, dramatically improving lesion conspicuity and anatomical localization. Whereas a standard chest X-ray integrates all tissue attenuation along the beam path into a single two-dimensional projection, a CT acquisition resolves individual axial, coronal, and sagittal planes with equal fidelity, enabling comprehensive three-dimensional anatomical analysis. This advantage is particularly pronounced for complex anatomical regions such as the chest, abdomen, pelvis, and spine. For foundational context on X-ray technology, refer to X-ray Machines: All You Need To Know As A Biomedical Engineer.
Compared to MRI, CT offers several practical advantages: significantly shorter acquisition times (seconds versus minutes), less sensitivity to patient motion, broader patient compatibility (including patients with pacemakers and metallic implants that are MRI-incompatible), and substantially lower cost per examination. CT is superior to MRI for evaluating osseous detail, acute hemorrhage, calcifications, and pulmonary parenchyma. However, MRI surpasses CT in soft-tissue contrast resolution for neurological, musculoskeletal, and pelvic pathology, and avoids ionizing radiation — an important consideration in pediatric and pregnant patients. The optimal imaging strategy is therefore lesion- and patient-specific, and biomedical engineers designing imaging pathways must understand these complementary strengths. For comparison with another major imaging modality, see From 1.5T to 7T and Beyond: How MRI Technology and AI are Reshaping Diagnostics.
What Are the Risks and Limitations of CT Scanners?
Ionizing Radiation Exposure and the ALARA Principle
The most clinically significant risk associated with CT imaging is exposure to ionizing radiation, which carries a theoretical stochastic risk of carcinogenesis proportional to cumulative dose. A standard abdominal-pelvic CT delivers an effective dose of approximately 8–15 millisieverts (mSv), compared to roughly 0.02 mSv for a chest X-ray and approximately 3 mSv for natural background radiation over one year. The radiation risk is of particular concern in pediatric patients, whose developing tissues are more radiosensitive and who have longer post-exposure lifetimes over which a radiation-induced malignancy could manifest. Epidemiological studies, including the landmark 2012 Pearce et al. study in The Lancet, have reported small but statistically significant increases in leukemia and brain tumor risk following CT exposure in childhood, reinforcing the imperative for dose optimization.
The guiding principle for managing CT radiation exposure is ALARA — As Low As Reasonably Achievable — which mandates that every examination be performed with the minimum dose necessary to produce diagnostically adequate images. Practical implementation of ALARA involves individualized dose modulation based on patient size and body habitus, selection of appropriate scan protocols (avoiding unnecessary multiphase acquisitions), utilization of iterative reconstruction algorithms to reduce tube current without sacrificing image quality, and strict clinical justification for every CT referral. Regulatory frameworks, including the FDA’s Initiative to Reduce Unnecessary Radiation Exposure from Medical Imaging (2010) and the Image Gently campaign for pediatric patients, have driven systemic improvements in CT dose management across healthcare institutions. Biomedical engineers responsible for CT commissioning and maintenance must be proficient in CT dosimetry tools including CT Dose Index (CTDI) and Dose Length Product (DLP) measurements.
Contrast Agent Reactions and Medical Contraindications
Iodinated contrast media (ICM) are administered intravenously in a large proportion of CT examinations to enhance vascular structures, lesion conspicuity, and organ perfusion patterns. While generally safe, contrast agents carry a defined risk profile that must be carefully managed. Adverse reactions range from mild (nausea, urticaria, warmth sensation) to moderate (bronchospasm, hypotension) and, rarely, severe anaphylactoid reactions that can be life-threatening. The reported incidence of severe reactions to modern low-osmolality non-ionic contrast agents is approximately 0.04%, but this risk is elevated in patients with prior contrast reactions, asthma, or significant atopy. Premedication protocols using corticosteroids and antihistamines are routinely employed in at-risk populations.
Contrast-induced acute kidney injury (CI-AKI) is a potentially serious complication in patients with pre-existing renal impairment, diabetes mellitus, or dehydration. Current evidence suggests that the risk of CI-AKI has been historically overestimated, particularly when modern low-osmolality agents are used with adequate hydration; however, caution remains warranted when estimated glomerular filtration rate (eGFR) falls below 30 mL/min/1.73m². Metformin, a commonly prescribed antidiabetic agent, must be withheld following contrast administration in renally compromised patients due to the risk of lactic acidosis. Additionally, CT scanning is contraindicated in pregnancy unless the clinical benefit clearly outweighs fetal radiation risk, particularly during the first trimester. Biomedical engineers designing CT workflow systems should incorporate electronic contraindication screening and automated contrast injection safety interlocks to reduce preventable adverse events.
Image Artifacts and Technical Limitations
CT images are susceptible to a variety of artifacts that can degrade image quality and potentially lead to diagnostic errors. Beam hardening artifacts arise because the polychromatic X-ray beam preferentially loses lower-energy photons as it traverses dense materials such as bone or metal implants, causing the remaining higher-energy beam to appear falsely attenuated. This manifests as dark streaking or cupping artifacts, most commonly seen around the petrous bones in head CT, the shoulders in chest CT, and adjacent to prosthetic joints or spinal fixation hardware. Manufacturers have developed beam-hardening correction algorithms and iterative metal artifact reduction (iMAR) techniques to mitigate these effects, though complete elimination remains challenging in heavily implanted patients.
Motion artifacts represent another significant challenge, particularly in cardiac, abdominal, and pediatric imaging where patient movement or physiological motion during data acquisition produces blurring, ghosting, or misregistration artifacts. Respiratory motion is addressed through breath-hold protocols or respiratory triggering, while cardiac motion necessitates electrocardiographic (ECG) gating — either prospective (triggering) or retrospective — to synchronize data acquisition with the cardiac cycle. Partial volume averaging, staircase artifacts in multiplanar reformatted images, and ring artifacts from detector miscalibration are additional technical limitations that quality assurance programs must systematically monitor. High cost, large equipment footprint, scanner-specific radiation shielding requirements, and limited access in low-resource healthcare settings further constrain the global deployment of CT technology, highlighting important equity considerations for healthcare systems engineering.
How Is the CT Scanner Evolving? Recent Innovations
AI-Powered Image Reconstruction and Deep Learning
Artificial intelligence, and deep learning in particular, is redefining the boundaries of CT image reconstruction and analysis. Traditional filtered back-projection (FBP) reconstruction, which has dominated CT since the 1970s, was progressively displaced by statistical iterative reconstruction (IR) algorithms in the 2010s, enabling significant dose reductions (30–60%) while maintaining diagnostic image quality. The current frontier is deep learning-based reconstruction (DLR), which trains convolutional neural networks on large datasets of paired high-dose and low-dose CT images to learn optimal noise suppression and spatial resolution characteristics. Commercial platforms such as Siemens Healthineers’ ADMIRE, GE HealthCare’s TrueFidelity, Canon Medical’s Advanced intelligent Clear-IQ Engine (AiCE), and Philips’ IMR represent the state of the art, each demonstrating superior noise texture and lesion detectability compared to conventional IR methods.
Beyond reconstruction, AI algorithms are being deployed for automated organ segmentation, lesion detection, triage prioritization, and quantitative imaging biomarker extraction. AI-powered CT triage tools for intracranial hemorrhage, pulmonary embolism, and aortic aneurysm have received FDA clearance and are demonstrably reducing time-to-diagnosis in emergency workflows. Radiomics — the high-throughput extraction of quantitative features from CT images — combined with machine learning models is enabling non-invasive tumor phenotyping, treatment response prediction, and survival outcome stratification. For biomedical engineers interested in the intersection of AI and medical imaging, the article Latest Advances of Artificial Intelligence in Healthcare in 2025 provides an excellent complementary resource, as does the Practical Roadmap for Biomedical Engineering Students to Learn Artificial Intelligence in Healthcare.
Photon-Counting CT Detectors: The Next Hardware Paradigm
Photon-counting detector CT (PCD-CT) represents the most significant hardware advancement in CT technology since the introduction of multi-slice systems. Unlike conventional energy-integrating detectors (EIDs), which sum the total charge deposited by all photons without discriminating their individual energies, photon-counting detectors register each individual photon and measure its energy level, assigning it to predefined energy bins. This fundamental difference confers multiple simultaneous advantages: elimination of electronic noise (since only photons above a set energy threshold are counted), improved spatial resolution through smaller detector pixel pitch, inherent spectral information acquisition at every scan (enabling material decomposition without the dose penalty of dual-source methods), and reduced metal artifacts.
The Siemens Healthineers NAEOTOM Alpha, which received FDA clearance in September 2021, was the world’s first commercially available whole-body PCD-CT scanner, utilizing cadmium telluride (CdTe) detector elements. Clinical studies have demonstrated that the NAEOTOM Alpha achieves submillimeter resolution (down to 0.2 mm) while simultaneously enabling multi-energy spectral imaging at standard radiation doses. GE HealthCare’s Revolution Apex platform represents another major advancement in the premium CT segment, incorporating advanced detector technology and AI reconstruction to push temporal and spatial resolution boundaries in cardiac and general imaging applications. These systems are particularly transformative for cardiovascular imaging, oncological staging, and musculoskeletal applications where fine structural detail is clinically determinative. The implications for biomedical engineers involved in CT procurement are substantial — PCD-CT systems require revised quality assurance protocols and infrastructure planning. For practical guidance, see the Buying Guide 2025: How to Choose the Right CT Scanner.
Spectral and Dual-Energy CT, Low-Dose Protocols, and Mobile CT
Dual-energy CT (DECT) and spectral CT have matured from research tools into established clinical technologies available across all major scanner platforms. By acquiring data at two different photon energy spectra — achieved via dual-source configurations, rapid kV-switching, dual-layer detector design (as in the Philips IQon Spectral CT), or split-filter techniques — DECT enables material decomposition, virtual monoenergetic image (VMI) generation, and quantitative tissue characterization beyond what is possible with conventional single-energy CT. Clinically validated applications include uric acid gout crystal detection, virtual non-contrast (VNC) imaging that eliminates the need for a separate unenhanced acquisition (reducing both dose and scan time), iodine map quantification for pulmonary embolism and hepatic lesion characterization, and virtual non-calcium (VNCa) maps for bone marrow edema assessment. The spectral data generated by these systems constitutes an emerging class of quantitative imaging biomarkers with significant implications for precision medicine pathways.
Low-dose CT protocols have been progressively refined through the combined application of iterative and deep learning reconstruction, automated tube current modulation (ATCM), organ-based dose modulation, and optimized scan geometry. The landmark National Lung Screening Trial (NLST), published in 2011, demonstrated a 20% reduction in lung cancer mortality with annual low-dose CT (LDCT) screening in high-risk smokers, leading to widespread adoption of lung cancer screening programs in the United States and Europe. Current LDCT protocols achieve effective doses below 1.5 mSv, approaching background radiation levels. In parallel, mobile and point-of-care CT systems — including the CereTom portable head CT (NeuroLogica, a Samsung company) and the BodyTom whole-body portable system — are extending CT diagnostic capability to intensive care units, stroke centers, and austere environments where patient transport is high-risk. The integration of CT with PET imaging in combined PET-CT systems provides co-registered anatomical and metabolic data in a single examination session, which has become the standard of care for oncological staging and treatment response assessment in many tumor types. For context on another important complementary imaging modality, see Ultrasound Imaging Systems Explained: Physical Principles Governing Acoustic Waves and Tissue Interaction.
Regulatory Standards and Manufacturer Ecosystem
CT scanners are Class II medical devices in the United States, subject to FDA 510(k) premarket notification requirements, and are regulated under 21 CFR Part 892. The primary international electrotechnical standard governing CT scanner safety and performance is IEC 60601-2-44 (Medical electrical equipment — Part 2-44: Particular requirements for the basic safety and essential performance of X-ray equipment for computed tomography), which specifies requirements for radiation output, mechanical safety, electromagnetic compatibility, and display performance. Manufacturers must also comply with ISO 13485 for quality management systems throughout the device lifecycle, from design and manufacturing through post-market surveillance. In the European Union, CT systems are regulated as Class IIb medical devices under the Medical Device Regulation (MDR 2017/745), requiring notified body conformity assessment and CE marking.
The global CT market is dominated by four major manufacturers: Siemens Healthineers (SOMATOM and NAEOTOM product families), GE HealthCare (Revolution CT family), Philips Healthcare (Incisive and IQon spectral CT), and Canon Medical Systems (Aquilion family). Each manufacturer offers a tiered product portfolio ranging from basic 16-slice systems for general radiology to premium photon-counting and dual-source platforms for advanced clinical and research applications. Secondary manufacturers, including Samsung NeuroLogica, United Imaging Healthcare, and Neusoft Medical, are expanding the competitive landscape, particularly in portable and value-segment markets. Biomedical engineers engaged in CT procurement should thoroughly evaluate application data packages (ADAPs), service contract terms, software upgrade pathways, and integration capabilities with existing RIS/PACS infrastructure. For a comprehensive overview of how medical devices are classified and regulated, refer to How Biomedical Devices Are Classified: Insights into FDA Classification, and for the broader regulatory landscape, see Key Organizations and Bodies in the Medical Device Field.
Key Takeaways and Tips for Biomedical Engineers
Engineering Considerations for CT Procurement, Installation, and Maintenance
The procurement and installation of a CT scanner represents one of the most complex capital equipment projects in a healthcare facility, requiring deep multidisciplinary collaboration between biomedical engineers, radiologists, radiation physicists, facilities management, and clinical informatics teams. Site planning must account for substantial structural requirements: scanner weight (typically 1,000–3,000 kg for the gantry assembly), floor load capacity, primary radiation shielding design (typically involving lead-lined walls and a controlled access zone), uninterruptible power supply (UPS) for high-voltage generator stability, chilled water cooling systems for X-ray tube heat dissipation, and dedicated high-bandwidth network infrastructure for DICOM data transmission to PACS. The room layout must comply with IEC 60601-1 general safety requirements and local building codes, and radiation shielding calculations must be performed by a certified medical physicist in accordance with NCRP Report No. 147 guidelines.
Preventive maintenance programs for CT scanners should be structured around manufacturer-specified service intervals and include systematic inspection of X-ray tube performance parameters (filament current, anode rotation speed, focal spot size), detector calibration (air calibration and offset correction), slip ring contact integrity, gantry mechanical balance and vibration analysis, table positioning accuracy, and laser alignment verification. X-ray tube replacement — typically required after 500,000–1,000,000 scan seconds or when tube output degrades below acceptable thresholds — is among the highest-cost maintenance events and should be factored into lifecycle cost modeling during procurement. Biomedical engineers should maintain detailed equipment history logs, track mean time between failures (MTBF) for key subsystems, and establish escalation protocols for scanner downtime to minimize impact on clinical throughput. Understanding the full scope of radiological device maintenance is a critical competency — Radiological Devices: All You Must Know as a Biomedical Engineer provides valuable foundational guidance.
Quality Assurance Protocols: CT Dosimetry and Phantom Testing
A rigorous CT quality assurance (QA) program is essential for ensuring consistent image quality, patient safety, and regulatory compliance. The American Association of Physicists in Medicine (AAPM) Task Group reports — particularly TG-233 on CT QA protocols — provide authoritative guidance on acceptance testing and ongoing performance monitoring. Core QA metrics include CT number accuracy and linearity (verified using tissue-equivalent phantom inserts with known HU values), spatial resolution (assessed via modulation transfer function and high-contrast bar pattern phantoms), low-contrast detectability (using low-contrast cylindrical targets in uniform background phantoms), image noise measurement, slice thickness verification, patient table incrementation accuracy, and gantry tilt accuracy. These tests should be performed at defined intervals: some daily, others weekly, monthly, or annually, as specified in the facility’s medical physics QA protocol.
CT dosimetry monitoring is mandated by regulatory bodies in most jurisdictions and should be performed using calibrated ionization chambers (pencil chambers) and standardized CTDI phantoms (16 cm head and 32 cm body) traceable to national measurement standards. The CTDIvol and DLP values displayed on the scanner console must be validated against independent measurements at commissioning and after major software updates. Dose reference levels (DRLs) — nationally or regionally established benchmarks representing the 75th percentile of dose distributions for standard examinations — provide a practical optimization framework: institutions consistently exceeding DRLs should investigate and remediate protocol settings. The ACR CT Accreditation Program provides a structured framework for QA documentation, phantom testing, and peer review that biomedical engineers can use to guide institutional compliance. Integration of CT dose monitoring software (such as Bayer Radimetrics, Sectra DoseTrack, or Philips DoseWise) into the imaging workflow enables automated dose tracking, outlier detection, and regulatory reporting at scale.
Career Paths, Emerging Roles, and Professional Development for CT Specialists
The rapid technological evolution of CT — encompassing AI reconstruction, photon-counting detectors, spectral imaging, and mobile platforms — is creating a growing demand for biomedical engineers with specialized CT expertise. Clinical application specialists employed by manufacturers require deep knowledge of both the physics and clinical utility of CT systems to support customer training and product optimization. Medical physics careers focused on CT dosimetry, protocol optimization, and QA program management are well-established and increasingly valued as dose awareness and regulatory scrutiny intensify. Healthcare technology management (HTM) professionals with CT-specific expertise command premium positioning in hospital biomedical engineering departments and independent service organizations (ISOs) that maintain CT fleets. Research engineers at academic medical centers and national laboratories are advancing next-generation detector technologies, AI reconstruction algorithms, and quantitative imaging biomarkers with direct translational impact.
Emerging roles are appearing at the intersection of CT imaging and data science, where engineers with combined competencies in signal processing, machine learning, and clinical imaging informatics are developing and validating AI-powered CT diagnostic tools. Regulatory affairs specialists with CT device expertise are needed to navigate the complex FDA 510(k) and De Novo pathways for AI/ML-enabled CT software as a medical device (SaMD). For biomedical engineers building their career trajectories, cultivating expertise in CT technology provides a durable competitive advantage given the modality’s central and irreplaceable role in modern healthcare. Broadening competency to include adjacent imaging modalities and device categories also strengthens a professional’s value — resources such as Top Skills Every Biomedical Engineer Should Master, Top Career Paths in Biomedical Engineering (2025), and Ethical Considerations in Biomedical Engineering: Balancing Innovation and Patient Safety offer valuable professional development context. Engineers should also consider developing expertise in complementary imaging modalities — for example, advanced ultrasound as explored in Advanced Ultrasound Technologies and Future Innovations in Medical Imaging — to become well-rounded imaging specialists capable of contributing across the full diagnostic imaging enterprise.
References
- Hounsfield, G. N. (1973). Computerized transverse axial scanning (tomography): Part 1. Description of system. The British Journal of Radiology, 46(552), 1016–1022. https://doi.org/10.1259/0007-1285-46-552-1016
- U.S. Food and Drug Administration. (2020). Computed Tomography (CT): Initiative to Reduce Unnecessary Radiation Exposure from Medical Imaging. FDA. https://www.fda.gov/radiation-emitting-products/medical-x-ray-imaging/computed-tomography-ct
- International Electrotechnical Commission. (2018). IEC 60601-2-44: Medical Electrical Equipment — Part 2-44: Particular Requirements for the Basic Safety and Essential Performance of X-ray Equipment for Computed Tomography (Edition 3.2). IEC. https://webstore.iec.ch/publication/63636
- Siemens Healthineers. (2021). NAEOTOM Alpha: The World’s First Photon-Counting CT Scanner. Siemens Healthineers. https://www.siemens-healthineers.com/computed-tomography/photon-counting-ct/naeotom-alpha
- GE HealthCare. (2023). Revolution CT Family: Clinical Performance at the Speed of Life. GE HealthCare Technologies. https://www.gehealthcare.com/products/computed-tomography/revolution-ct
- American Association of Physicists in Medicine. (2019). AAPM Task Group 233: Performance Evaluation of Computed Tomography Systems. AAPM Report TG-233. American Association of Physicists in Medicine. https://www.aapm.org/pubs/reports/RPT_233.pdf
- Willemink, M. J., Noël, P. B., & Fleischmann, D. (2019). The evolution of image reconstruction for CT — from filtered back projection to artificial intelligence. European Radiology, 29(5), 2185–2195. https://doi.org/10.1007/s00330-018-5810-7
- McCollough, C. H., Leng, S., Yu, L., & Fletcher, J. G. (2015). Dual- and multi-energy CT: principles, technical approaches, and clinical applications. Radiology, 276(3), 637–653. https://doi.org/10.1148/radiol.2015142631
- Pearce, M. S., Salotti, J. A., Little, M. P., McHugh, K., Lee, C., Kim, K. P., … & Berrington de González, A. (2012). Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. The Lancet, 380(9840), 499–505. https://doi.org/10.1016/S0140-6736(12)60815-0
- National Lung Screening Trial Research Team. (2011). Reduced lung-cancer mortality with low-dose computed tomographic screening. New England Journal of Medicine, 365(5), 395–409. https://doi.org/10.1056/NEJMoa1102873