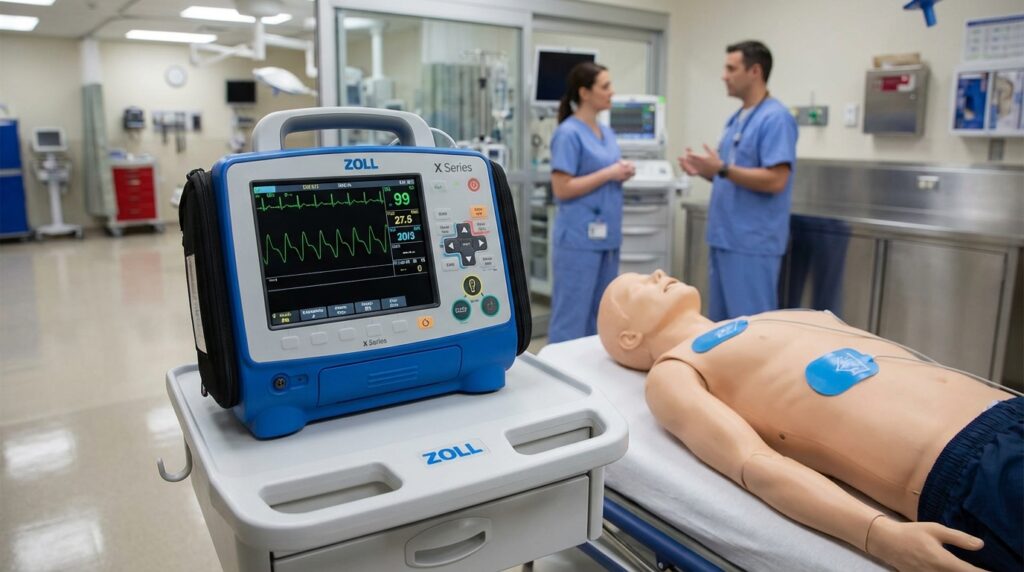
A cardiac monitor is one of the most critical instruments in modern clinical settings, providing real-time, continuous surveillance of a patient’s heart rhythm, ECG waveforms, and associated vital signs. For biomedical engineers, it represents a sophisticated convergence of biopotential sensing, analog front-end electronics, digital signal processing, alarm management systems, and wireless communication — all engineered to detect life-threatening arrhythmias at the earliest possible moment. This comprehensive overview covers the device’s history, operating principles, component architecture, clinical variants, regulatory landscape, recent AI-driven innovations, and essential guidance for engineers who design, procure, maintain, or troubleshoot these systems.
- What is a Cardiac Monitor?
- Why is a Cardiac Monitor Used?
- How Does a Cardiac Monitor Work?
- What Are the Main Components of a Cardiac Monitor?
- What Types and Variants of Cardiac Monitors Exist?
- What Are the Main Benefits of a Cardiac Monitor?
- What Are the General Risks and Limitations?
- How Is the Cardiac Monitor Evolving? Recent Innovations
- Key Takeaways and Tips for Biomedical Engineers
What is a Cardiac Monitor?
A cardiac monitor is a sophisticated medical device designed to continuously observe, display, and record a patient’s cardiac electrical activity and associated vital signs in real time. Unlike a standard Electrocardiograph (ECG), which captures a snapshot in time, cardiac monitors provide sustained surveillance — making them indispensable in intensive care units (ICUs), emergency departments, operating theaters, and step-down units. For biomedical engineers, understanding these devices is foundational within the broader landscape of cardiovascular devices.
Bedside Cardiac Monitor vs. Telemetry Monitor
A bedside cardiac monitor is a stationary unit placed at the patient’s bedside, physically tethered via lead wires. It continuously displays ECG waveforms, heart rate, SpO₂, blood pressure, respiratory rate, and temperature on a dedicated screen. In contrast, a telemetry monitor employs wireless radiofrequency (RF) transmission — typically operating in the 608–614 MHz or 1.4 GHz WMTS bands — allowing patients to ambulate freely while their cardiac rhythm is observed remotely at a central nursing station. Both categories are classified as Class II medical devices under FDA regulations, as detailed in resources covering how biomedical devices are classified.
Continuous ECG Recording and Vital Sign Monitoring
Modern cardiac monitors go far beyond simple ECG display. They integrate multiparameter monitoring capabilities — capturing invasive and non-invasive blood pressure (IBP/NIBP), pulse oximetry (SpO₂), end-tidal CO₂ (EtCO₂), core temperature, and cardiac output. Continuous ECG recording enables automated arrhythmia detection, ST-segment analysis, and QTc interval trending, all of which feed into intelligent alarm management systems. This integration places cardiac monitors within the larger family of biomedical devices that form the backbone of patient safety infrastructure.
Historical Evolution: From Einthoven to Modern ICU Monitors
The story of cardiac monitoring begins with Willem Einthoven, who in 1902 recorded the first human ECG using a string galvanometer, earning the Nobel Prize in Physiology or Medicine in 1924. The introduction of the bipolar limb leads — Lead I, II, and III — established the foundational Einthoven triangle still referenced today. In the 1950s and 1960s, cathode ray oscilloscopes enabled continuous bedside display, giving rise to the first coronary care units (CCUs). The transition from analog to digital signal processing in the 1980s and 1990s transformed monitors into fully programmable, networked devices. Today’s multiparameter ICU monitors, integrated with hospital information systems (HIS) and picture archiving systems akin to those described in a comprehensive guide to PACS for biomedical engineers, represent the culmination of over a century of innovation.
Why is a Cardiac Monitor Used?
The primary rationale for cardiac monitoring is patient safety through early detection of life-threatening arrhythmias and hemodynamic instability. Just as imaging devices such as CT scanners provide anatomical data, cardiac monitors deliver continuous physiological data streams that guide clinical decision-making.
Arrhythmia Detection and Management
Cardiac monitors are the primary tool for identifying arrhythmias such as ventricular fibrillation (VF), ventricular tachycardia (VT), atrial fibrillation (AF), complete heart block, and bradyarrhythmias. Automated QRS detection algorithms classify each beat in real time, triggering immediate alarms that prompt clinical intervention. This is especially critical in post-operative patients, those recovering from myocardial infarction, and critically ill patients in the ICU. The fetal cardiac monitor represents a specialized variant of this technology applied to obstetric care.
Perioperative and Critical Care Monitoring
In operative settings and ICUs, cardiac monitors integrate with infusion systems — including infusion pumps — ventilators, and electronic health records (EHRs) to provide a holistic view of patient status. ST-segment monitoring helps detect myocardial ischemia in real time, while continuous blood pressure trending alerts clinicians to hemodynamic compromise. Biomedical engineers must understand these clinical use cases to properly configure alarm thresholds and maintain device integrity.
Ambulatory and Remote Monitoring
Beyond acute care, cardiac monitors extend into ambulatory settings through Holter monitors and wearable devices that capture rhythm data over 24–48 hours or longer. Remote telemetry enables monitoring of patients in general wards or even at home, aligning with the growing trend of patient-centered care. This intersects with the ethical considerations in biomedical engineering around data privacy, informed consent, and equitable access to monitoring technology.
How Does a Cardiac Monitor Work?
The operation of a cardiac monitor involves a precisely coordinated chain of biophysical sensing, analog signal conditioning, digitization, digital signal processing, and intelligent display with alarm generation. Understanding this signal chain is one of the top skills every biomedical engineer should master.
Biopotential Sensing and ECG Lead Systems
Cardiac electrical activity — generated by depolarization and repolarization of myocardial cells — produces biopotentials on the body surface in the range of 0.5–5 mV with a frequency bandwidth of 0.05–150 Hz. Silver/silver chloride (Ag/AgCl) electrodes placed on the skin transduce these ionic potentials into electronic signals. Depending on the clinical requirement, 3-lead configurations (RA, LA, LL) provide basic rhythm monitoring; 5-lead systems add a chest lead (V1) enabling ST-segment analysis; and 12-lead configurations deliver the full diagnostic ECG used in myocardial infarction assessment. This electrochemical transduction principle is analogous to the piezoelectric transduction employed in ultrasound imaging systems.
Analog Front-End Signal Conditioning
The raw biopotential is extraordinarily small and susceptible to interference. An instrumentation amplifier (INA) with a high common-mode rejection ratio (CMRR ≥ 80 dB) amplifies the differential signal while rejecting common-mode noise — including 50/60 Hz powerline interference. A notch filter (50 or 60 Hz) further attenuates powerline artifacts. A band-pass filter (typically 0.05–40 Hz for monitoring mode, 0.05–150 Hz for diagnostic mode) removes baseline wander and high-frequency muscle artifact. These filtering strategies are critical design parameters that biomedical engineers must optimize for each clinical application.
Analog-to-Digital Conversion and Digital Signal Processing
The conditioned analog ECG signal is digitized by an analog-to-digital converter (ADC) at a sampling rate of 500–1000 samples per second with 12–16 bit resolution, ensuring accurate representation of rapid QRS complexes (duration ~80–120 ms). The digitized signal is processed by a microprocessor or digital signal processor (DSP) running QRS detection algorithms — most notably adaptations of the Pan-Tompkins algorithm — which differentiate true QRS complexes from noise and artifacts. Derived parameters such as heart rate, RR intervals, QTc, and ST-level deviations are computed in real time. The microprocessor also manages alarm logic, display rendering, data storage, and network communication. This computational architecture parallels the image reconstruction pipelines found in SPECT imaging systems.
Alarm Algorithms and Clinical Decision Support
Modern cardiac monitors employ layered alarm algorithms that classify rhythms as normal, advisory, or critical. Ventricular fibrillation and asystole trigger the highest-priority alarms with immediate audio-visual alerts and nurse call relay activation. ST-segment alarms are time-gated to avoid false positives from patient movement. Advanced monitors incorporate artificial intelligence for adaptive alarm thresholding, significantly reducing alarm fatigue — a major patient safety concern identified by regulatory bodies such as those outlined in resources covering key organizations in the medical device field.
What Are the Main Components of a Cardiac Monitor?
A cardiac monitor is an integrated system comprising hardware and software subsystems that must function reliably in demanding clinical environments. Biomedical engineers engaged in procurement, maintenance, or design must have thorough knowledge of each component, as outlined in comprehensive guides such as why anatomy and physiology knowledge matters for biomedical engineers.
Electrodes and Lead Wires
Disposable Ag/AgCl snap electrodes are the clinical standard, offering low and stable electrode-skin interface impedance (typically <5 kΩ after preparation). The silver/silver chloride chemistry minimizes DC offset potentials, which could saturate amplifier stages. Lead wires are shielded, typically color-coded per IEC 60601-2-27 (red, yellow, green, black, white for 5-lead) and terminated in snap connectors. Lead wire integrity — including resistance, shielding continuity, and insulation — is a common failure point requiring regular inspection as part of a preventive maintenance program.
Patient Module and Analog Front-End
The patient module consolidates all physiological measurement interfaces. The analog front-end (AFE) comprises the INA, protection circuitry (to survive defibrillation pulses of up to 5 kV), right-leg drive (RLD) circuit for active common-mode rejection, and the filter chain described in Section 3. Modular patient interfaces allow rapid replacement in the field — a significant serviceability advantage biomedical engineers should factor into total cost of ownership assessments.
Display, Microprocessor, and Data Management
Contemporary monitors feature high-resolution TFT LCD or OLED touchscreens (typically 12–21 inches diagonal) rendering multiple simultaneous waveform channels at 25 mm/s sweep speed. The central microprocessor or system-on-chip (SoC) runs a real-time operating system (RTOS) ensuring deterministic alarm response times (<1 second). Onboard storage — flash memory or solid-state drives — archives waveform data for retrospective review, trend analysis, and regulatory compliance. Network connectivity via IEEE 802.11 Wi-Fi or wired Ethernet enables HL7-compliant data export to EHR systems, analogous to DICOM-based image management discussed in the PACS guide.
Battery Backup, Wireless Telemetry Module, and Nurse Call Relay
Lithium-ion battery systems provide 4–8 hours of operation during patient transport, meeting IEC 60601-1 electrical safety standards. The wireless telemetry module — operating in the Medical Device Radiocommunications Service (MedRadio) or ISM bands — transmits compressed ECG data to central stations with latency under 500 ms. The nurse call relay interface (typically a dry-contact output) integrates the monitor’s alarm system with hospital-wide nurse call infrastructure, ensuring that critical arrhythmia alerts propagate to remote caregivers. Cybersecurity of wireless data links is increasingly scrutinized, paralleling concerns discussed in the context of medical imaging network security.
What Types and Variants of Cardiac Monitors Exist?
The cardiac monitor market encompasses a diverse array of device types, each optimized for specific clinical environments and patient populations. Understanding this taxonomy is essential for biomedical engineers involved in equipment planning, as outlined in guides to top career paths in biomedical engineering.
Bedside and Central Station Monitors
Bedside monitors are the workhorse of ICU and high-acuity care. They support 5- and 12-lead ECG, invasive hemodynamic monitoring, capnography, and advanced respiratory parameters. Examples include the Philips IntelliVue MX800 and GE Healthcare CARESCAPE B850. Central station monitors aggregate waveform data from multiple bedside units — often 16–64 beds — at a nursing station, enabling a single clinician to surveil an entire unit. These systems run sophisticated arrhythmia analysis engines and maintain detailed trend archives. Their architecture is conceptually similar to the centralized image management approach of PACS systems in radiology.
Telemetry and Portable/Transport Monitors
Telemetry monitors use wireless transmission to enable monitoring of ambulatory patients on general wards. They typically support 3- or 5-lead ECG and transmit to a central receiving station. Portable or transport monitors are ruggedized, battery-powered units designed for patient transport between departments — for example, from the ED to the ICU or from the ward to imaging suites such as those housing MRI systems. They must be MRI-compatible or clearly labeled as contraindicated in the MRI environment.
Wearable and Ambulatory Cardiac Monitors
Wearable cardiac monitors — including patch-based monitors (e.g., iRhythm Zio Patch), smart garments, and implantable loop recorders — extend monitoring to days, weeks, or even years. These devices leverage miniaturized AFEs, Bluetooth Low Energy (BLE) transmission, and cloud-based AI analysis platforms. They represent the frontier of medical device innovation, offering population-level cardiac screening capabilities. Biomedical engineers developing or evaluating these devices must consider battery longevity, skin biocompatibility, data security, and the AI roadmap for healthcare applications.
Comparison of Cardiac Monitor Types
| Type | Leads | Display Size | Connectivity | Primary Use | Example Models |
|---|---|---|---|---|---|
| Bedside Monitor | 3, 5, 12-lead | 12–21 inch TFT/OLED | Wired LAN, Wi-Fi, HL7 | ICU, CCU, OR | Philips IntelliVue MX800, GE CARESCAPE B850 |
| Central Station Monitor | Aggregated from bedside | Multiple large displays | Wired LAN, EHR integration | Nursing station surveillance | Philips IntelliCenter, GE CIC Pro |
| Telemetry Monitor | 3 or 5-lead | Remote display at station | RF wireless (608–614 MHz / 1.4 GHz) | General ward, step-down unit | Philips Intellivue MX40, Spacelabs Ultraview |
| Portable/Transport Monitor | 3 or 5-lead | 5–10 inch, ruggedized | Wi-Fi, Bluetooth, LAN | Intrahospital transport | Mindray iMEC8, Philips SureSigns VS4 |
| Multiparameter Monitor | 5 or 12-lead | 10–15 inch touchscreen | Wi-Fi, wired LAN, HL7/DICOM | ICU, ED, high-dependency units | Drager Infinity Delta, Nihon Kohden BSM-3500 |
| Wearable Cardiac Monitor | 1–2 lead (patch) | No onboard display | Bluetooth, cellular, cloud | Ambulatory, remote patient monitoring | iRhythm Zio Patch, BioTelemetry MCOT |
Selecting the appropriate cardiac monitor type requires careful analysis of clinical workflows, patient acuity, facility infrastructure, and regulatory compliance requirements — competencies that define excellence in the biomedical engineering profession. As with other complex medical systems such as CT scanners and ultrasound machines, cardiac monitors demand a multidisciplinary understanding of electronics, physiology, clinical informatics, and patient safety engineering.
What Are the Main Benefits of a Cardiac Monitor?
Cardiac monitors deliver measurable, evidence-based clinical advantages that make them indispensable across ICUs, CCUs, step-down units, and operating rooms. For biomedical engineers working with cardiovascular devices, understanding these benefits is essential for procurement decisions, preventive maintenance prioritization, and technology assessment.
Early Arrhythmia Detection and Continuous ST-Segment Analysis
The primary clinical value of a cardiac monitor lies in its ability to detect life-threatening arrhythmias—ventricular fibrillation, ventricular tachycardia, complete heart block, and atrial fibrillation—within seconds of onset. Modern monitors perform continuous ST-segment analysis across multiple leads, alerting clinicians to evolving myocardial ischemia before irreversible infarction occurs. Studies in CCU settings consistently demonstrate reductions in cardiac mortality of 15–25% attributable to real-time continuous monitoring and rapid response protocols enabled by these systems. The electrocardiograph (ECG) forms the foundation of this capability, and multiparameter monitors extend it with additional physiological context.
Non-Invasive Vital Signs Integration
Contemporary cardiac monitors integrate SpO2 pulse oximetry, non-invasive blood pressure (NIBP), end-tidal CO2 (EtCO2), and skin/core temperature into a single unified display. This multiparameter approach reduces patient cable burden, simplifies clinical workflow, and enables correlation of hemodynamic trends. EtCO2 monitoring is particularly valuable during procedural sedation and post-extubation surveillance, providing a non-invasive surrogate for ventilatory adequacy. Integration with infusion pump data streams via interoperability buses further enriches the clinical picture available at bedside and central stations.
Centralized Monitoring and Nurse Station Integration
Central monitoring stations aggregate waveform data from 16 to 128 beds simultaneously, enabling a dedicated monitoring technician to surveil an entire unit. Networked architectures transmit data over hospital Ethernet or Wi-Fi, feeding central displays with real-time ECG strips, trend graphs, and alarm queues. This architecture mirrors the centralized data management philosophy seen in PACS systems for biomedical engineers, where centralized data access improves response time and reduces missed critical events. Centralized monitoring has been shown to decrease response times to arrhythmia events by up to 40% in large academic medical centers.
Fetal and Specialized Monitoring Extensions
Cardiac monitoring platforms extend beyond adult care into obstetrics, where fetal cardiac monitors provide continuous cardiotocography. The same signal acquisition and alarm management principles apply, underscoring the versatility of the underlying biomedical engineering framework across patient populations.
What Are the General Risks and Limitations?
No medical device is without risk. Cardiac monitors present several technical, physiological, and operational challenges that biomedical engineers must understand to support safe deployment. These risks span hardware vulnerabilities, signal integrity issues, and systemic human factors problems. Understanding ethical considerations in balancing innovation and patient safety is equally important when managing these risks in practice.
Alarm Fatigue and False Alarm Burden
Published clinical literature reports false alarm rates of 85–99% in ICU environments, meaning the overwhelming majority of monitor alarms do not represent true clinical emergencies. This phenomenon—alarm fatigue—leads nursing staff to disable or silence alarms, creating genuine patient safety hazards. The Joint Commission has repeatedly flagged alarm management as a National Patient Safety Goal. Biomedical engineers must collaborate with clinical informatics teams to optimize alarm threshold settings, implement alarm escalation hierarchies, and use AI-based filtering to suppress artifact-driven false positives.
Motion Artifact, EMG Interference, and Electrode Skin Reactions
Motion artifact from patient movement, shivering, or physical therapy sessions degrades ECG signal quality and triggers spurious alarms. Electromyographic (EMG) noise from skeletal muscle activity can mimic arrhythmic waveforms in automated detection algorithms. Additionally, prolonged electrode contact causes contact dermatitis in 10–15% of monitored patients, particularly with latex-containing adhesives or patients with sensitive skin. Lead displacement—a common occurrence during patient repositioning or transport—is one of the most frequent causes of monitoring interruption. Biomedical engineers should specify hydrogel electrodes with hypoallergenic adhesives and implement lead-off detection algorithms with appropriate alert escalation.
Electromagnetic Interference and EMC Susceptibility
Cardiac monitors operating in environments with high-power equipment—electrosurgical units, MRI scanners, therapeutic ultrasound—face electromagnetic interference (EMI) that can corrupt signal acquisition or cause device malfunction. IEC 60601-1-2 (EMC standard) mandates immunity testing, but real-world hospital environments often exceed test conditions. Biomedical engineers should conduct EMC site surveys prior to monitor deployment, particularly in hybrid OR and interventional cardiology suites. The EMI management challenges in cardiac monitoring parallel those encountered in CT scanner environments and other high-energy diagnostic modalities.
Wireless Telemetry Data Security Vulnerabilities
Wireless cardiac telemetry systems transmit protected health information (PHI) over hospital Wi-Fi and proprietary RF bands. These systems are susceptible to man-in-the-middle attacks, denial-of-service disruptions, and unauthorized data interception if not properly hardened. The FDA’s 2023 cybersecurity guidance for networked medical devices applies directly to telemetry monitors. Biomedical engineers must ensure TLS 1.3 encryption, certificate-based authentication, and network segmentation via VLANs as baseline security controls—aligning with broader FDA device classification and regulatory requirements.
How Is the Cardiac Monitor Evolving? Recent Innovations
The cardiac monitoring landscape is undergoing rapid transformation driven by artificial intelligence, miniaturization, wireless connectivity, and health system interoperability. Biomedical engineers positioned at the intersection of clinical engineering and digital health must track these developments closely, just as the field monitors the latest AI advances in healthcare and innovations in imaging such as AI-driven MRI technology.
AI-Powered Arrhythmia Detection and Predictive Analytics
Deep learning convolutional neural networks (CNNs) trained on millions of annotated ECG samples now achieve cardiologist-level accuracy in detecting atrial fibrillation, STEMI patterns, and even subclinical left ventricular dysfunction from standard 12-lead recordings. Platforms such as the FDA-cleared ECG AI from Mayo Clinic and algorithms integrated into Philips IntelliVue and GE Healthcare MUSE systems represent the current state of the art. Predictive deterioration algorithms analyze multiparameter trend data to generate early warning scores (EWS), alerting rapid response teams up to 6 hours before a patient decompensates. For biomedical engineers pursuing these skills, a practical roadmap for learning AI in healthcare provides structured guidance.
Wearable and Patch-Based 12-Lead Monitoring
Miniaturization has enabled hospital-grade ECG monitoring in consumer and clinical wearable form factors. AliveCor KardiaMobile 6L delivers a 6-lead ECG via a smartphone-attached electrode pad, while the Apple Watch Series 9 provides single-lead ECG with FDA-cleared AF detection. Clinical-grade patch monitors such as iRhythm Zio XT record continuous single-lead ECG for up to 14 days, enabling ambulatory arrhythmia diagnosis without traditional Holter recorders. These devices comply with IEC 60601-2-47 (ambulatory ECG) and are transforming outpatient cardiac surveillance. This miniaturization trend parallels advances seen in advanced ultrasound technologies and portable imaging.
Wireless Telemetry: Wi-Fi 6 and Bluetooth 5.0 Platforms
Next-generation wireless cardiac telemetry systems leverage Wi-Fi 6 (802.11ax) for high-density, low-latency data transmission in crowded hospital RF environments, and Bluetooth 5.0 for short-range wearable sensor connectivity. Wi-Fi 6’s OFDMA scheduling reduces contention in environments with hundreds of connected devices, improving telemetry reliability. IEC 60601-2-49 specifically addresses multiparameter patient monitoring telemetry performance requirements, including alarm latency and data integrity specifications that biomedical engineers must verify during acceptance testing.
Cloud Connectivity, EHR Integration, and Remote Monitoring
Cloud-connected cardiac monitoring platforms enable cardiologists to review ECG strips, trend data, and alarm histories from any location via HIPAA-compliant web portals. HL7 FHIR-based APIs facilitate bidirectional integration with Epic, Cerner, and Oracle Health EHR systems, automatically populating flowsheets with vital signs and arrhythmia annotations. Remote patient monitoring (RPM) programs for post-discharge heart failure and AF patients use cloud-connected wearables to reduce 30-day readmission rates. This interoperability architecture shares design principles with PACS/DICOM ecosystem integration strategies that biomedical engineers already manage.
Key Takeaways and Tips for Biomedical Engineers
Effective stewardship of cardiac monitoring infrastructure requires biomedical engineers to combine technical expertise with clinical collaboration and regulatory awareness. The top skills every biomedical engineer should master include device management competencies directly applicable to cardiac monitoring programs. For broader context on device categories, the ultimate guide to biomedical devices provides an excellent reference framework.
Calibration, Preventive Maintenance, and Lead Placement Verification
Establish a preventive maintenance (PM) schedule aligned with manufacturer specifications and AAMI EC38 recommendations, including annual amplitude accuracy verification (±5% tolerance per AAMI EC11), frequency response testing (0.05–150 Hz for diagnostic quality), and CMRR measurement. Lead placement verification should be part of nursing competency programs co-developed with biomedical engineering. Electrode lot traceability and rotation protocols (every 24–48 hours for ICU patients) reduce skin reaction incidence and maintain signal quality. Battery management for portable and telemetry units must include capacity testing under load conditions, not merely voltage checks, to prevent unexpected mid-shift failures.
Alarm Customization and Fatigue Mitigation Strategies
Work with clinical staff to implement unit-specific alarm parameter sets rather than factory defaults. Use evidence-based alarm limits from published literature (e.g., HR alarm ranges of 40–130 bpm for general wards versus 30–150 bpm for ICU) and activate delay-before-alarm features (typically 5–15 seconds) to suppress self-terminating arrhythmias. Audit alarm data quarterly using monitor event logs to identify high-alarm-burden devices and beds. Collaborate with nursing informatics on alarm escalation middleware solutions such as Vocera or Ascom nurse call integration. Understanding key regulatory organizations in the medical device field helps contextualize compliance requirements for alarm management.
EMC Site Surveys and Cybersecurity Hardening
Conduct RF spectrum surveys (100 kHz–6 GHz) before deploying wireless telemetry in new clinical areas, documenting existing interference sources and establishing acceptable noise floors per IEC 60601-1-2 immunity levels. For cybersecurity, follow the FDA’s 2023 Cybersecurity in Medical Devices guidance: change default credentials immediately upon deployment, disable unused network ports, implement role-based access control (RBAC), apply manufacturer security patches within 30 days of release, and document all remote access sessions. Network segmentation isolates monitoring systems from general hospital IT traffic, limiting breach propagation pathways. These principles align with cybersecurity best practices for radiological devices.
Regulatory Compliance, Standards Adherence, and Career Development
Cardiac monitors must comply with IEC 60601-1 (general electrical safety), IEC 60601-2-27 (ECG monitoring performance), IEC 60601-2-49 (multiparameter monitoring with telemetry), IEC 60601-1-2 (EMC), ISO 13485 (quality management), and AAMI EC11/EC38/EC57 ECG standards. FDA 510(k) premarket clearance is required for all new monitor models entering the US market. Maintain a device inventory with UDI numbers, software version tracking, and PM completion records to support Joint Commission surveys. Biomedical engineers pursuing specialization in cardiac monitoring will find that a career in clinical technology management offers significant growth opportunities as AI-integrated monitoring platforms proliferate.
References
- IEC 60601-1:2005+AMD1:2012+AMD2:2020 – Medical Electrical Equipment – Part 1: General Requirements for Basic Safety and Essential Performance. International Electrotechnical Commission. https://www.iec.ch
- IEC 60601-2-27:2011+AMD1:2018 – Particular Requirements for the Basic Safety and Essential Performance of Electrocardiographic Monitoring Equipment. International Electrotechnical Commission. https://www.iec.ch
- IEC 60601-2-49:2011+AMD1:2017 – Particular Requirements for the Basic Safety and Essential Performance of Multifunction Patient Monitoring Equipment. International Electrotechnical Commission. https://www.iec.ch
- IEC 60601-1-2:2014+AMD1:2020 – General Requirements for Basic Safety and Essential Performance – Collateral Standard: Electromagnetic Disturbances. International Electrotechnical Commission. https://www.iec.ch
- AAMI EC11:2012/(R)2022 – Diagnostic Electrocardiographic Devices. Association for the Advancement of Medical Instrumentation. https://www.aami.org
- AAMI EC38:2007/(R)2014 – Ambulatory Electrocardiographs. Association for the Advancement of Medical Instrumentation. https://www.aami.org
- AAMI EC57:2012/(R)2020 – Testing and Reporting Performance Results of Cardiac Rhythm and ST-Segment Measurement Algorithms. Association for the Advancement of Medical Instrumentation. https://www.aami.org
- ISO 13485:2016 – Medical Devices – Quality Management Systems – Requirements for Regulatory Purposes. International Organization for Standardization. https://www.iso.org
- U.S. Food and Drug Administration. (2023). Cybersecurity in Medical Devices: Quality System Considerations and Content of Premarket Submissions. FDA Guidance Document. https://www.fda.gov
- U.S. Food and Drug Administration. (2022). 510(k) Premarket Notification – Cardiac Monitor Guidance. FDA. https://www.fda.gov
- Philips Healthcare. (2024). IntelliVue Patient Monitoring System – Technical and Clinical Reference Guide. Philips Medical Systems. https://www.philips.com/healthcare
- GE Healthcare. (2024). CARESCAPE Monitor B Series – Clinical Reference Manual. GE HealthCare Technologies. https://www.gehealthcare.com
- Mindray Medical International. (2023). BeneVision N Series Patient Monitor – Service and Technical Manual. Mindray. https://www.mindray.com
- Nihon Kohden Corporation. (2023). BSM-9100 Series Life Scope G Patient Monitor – Operator’s Manual. Nihon Kohden. https://www.nihonkohden.com
- Drew, B. J., et al. (2004). Practice Standards for Electrocardiographic Monitoring in Hospital Settings: An American Heart Association Scientific Statement. Circulation, 110(17), 2721–2746. https://doi.org/10.1161/01.CIR.0000145144.56673.59
- Cvach, M. (2012). Monitor Alarm Fatigue: An Integrative Review. Biomedical Instrumentation & Technology, 46(4), 268–277. https://doi.org/10.2345/0899-8205-46.4.268
- Hannun, A. Y., et al. (2019). Cardiologist-Level Arrhythmia Detection and Classification in Ambulatory Electrocardiograms Using a Deep Neural Network. Nature Medicine, 25(1), 65–69. https://doi.org/10.1038/s41591-018-0268-3
- The Joint Commission. (2023). National Patient Safety Goals Effective January 2024: NPSG.06.01.01 – Alarm Management. The Joint Commission. https://www.jointcommission.org
- Biomedevices.com. What Are Cardiovascular Devices: All You Need to Know as a Biomedical Engineer. https://biomedevices.com/what-are-cardiovascular-devices-all-you-need-to-know-as-a-biomedical-engineer/
- Biomedevices.com. Electrocardiograph (ECG). https://biomedevices.com/electrocardiograph-ecg/
- Biomedevices.com. Fetal Cardiac Monitor. https://biomedevices.com/fetal-cardiac-monitor/